Free Online Help: Given the following delta H values H2+1/2O2--->H2O delta H =-285.8 H2O2---->H2+O2 delta H = 187.6 Calculate delta H rxn for the following reaction H2O2--->H2O + 1/2O2

SOLVED: The enthalpy changes, 4 H , for three reactions are given. Hz(g) + Zo,t) 3 HzO() AH -286 kJlmol Ca(s) + 2Ht(aq) Cal+(aq) Hz(g) AH -544 kllmol CaO(s) + 2Ht(aq) Calt(aq) +





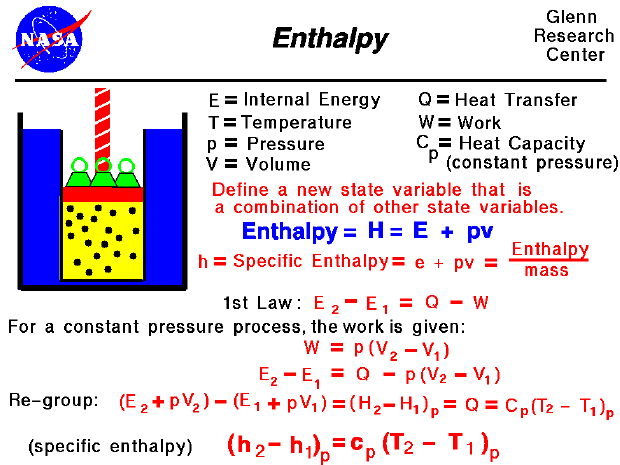
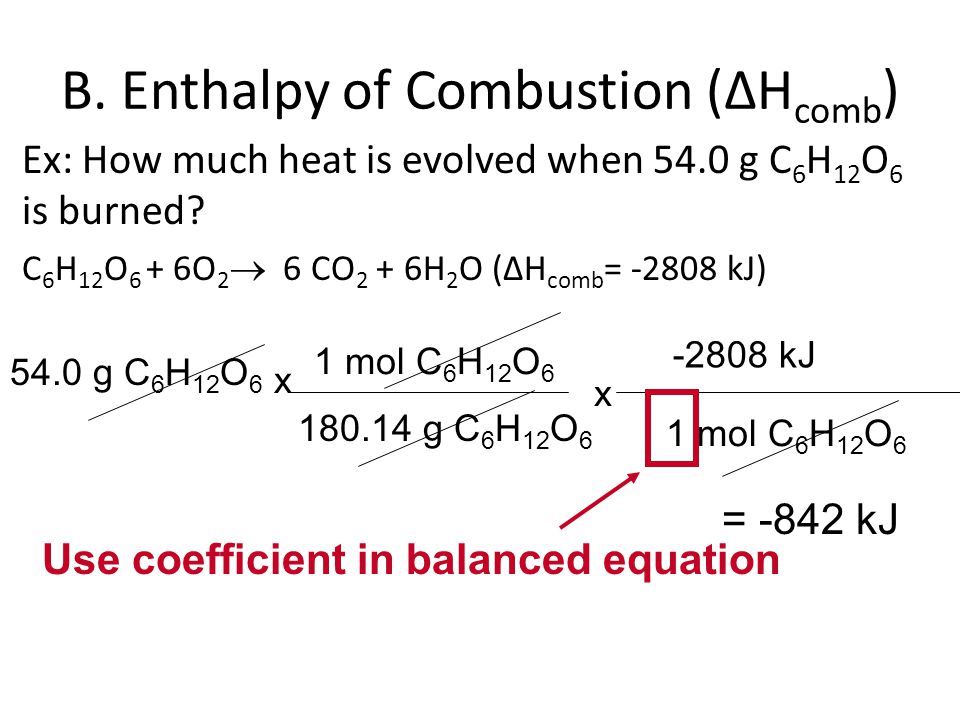



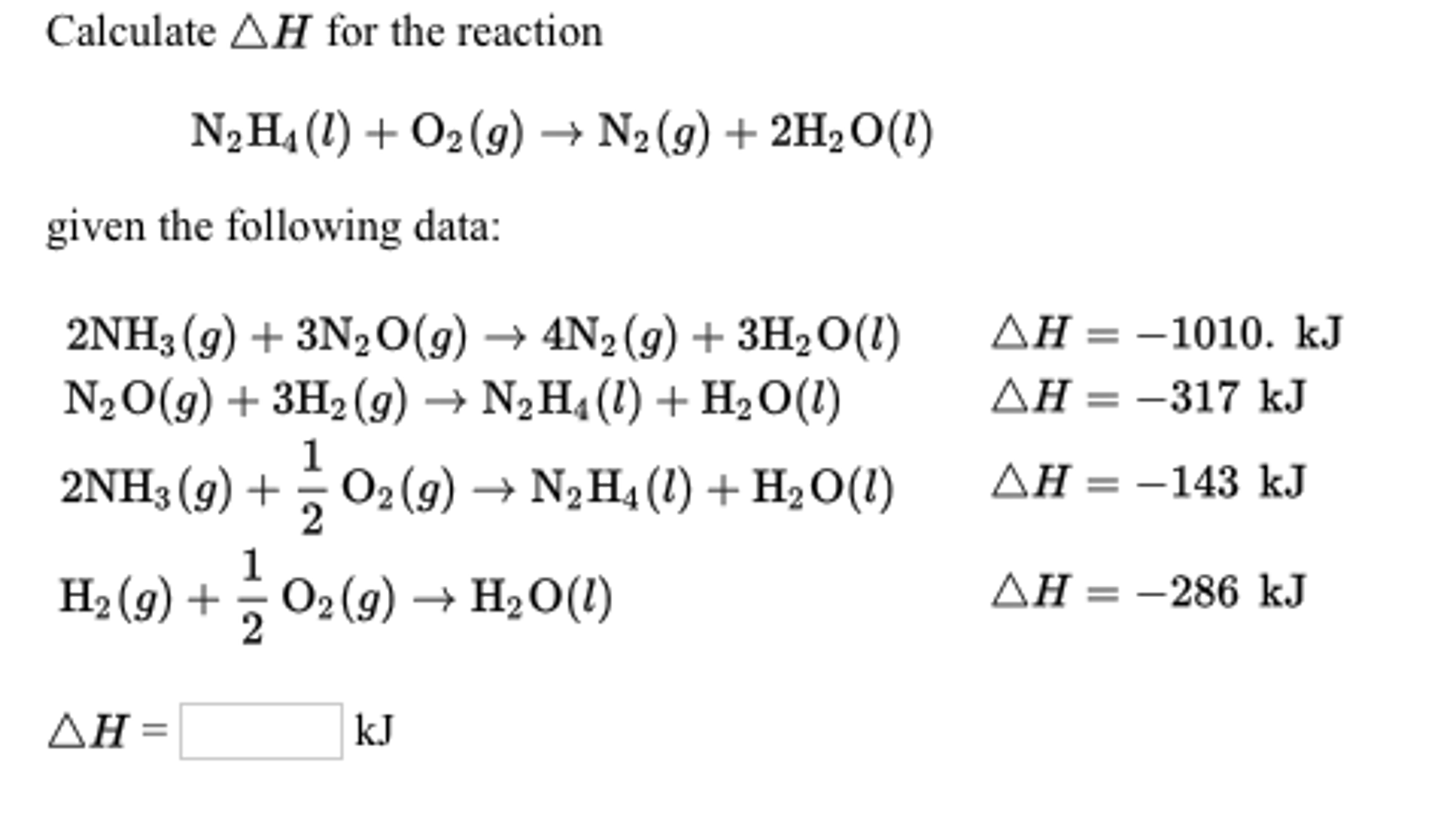

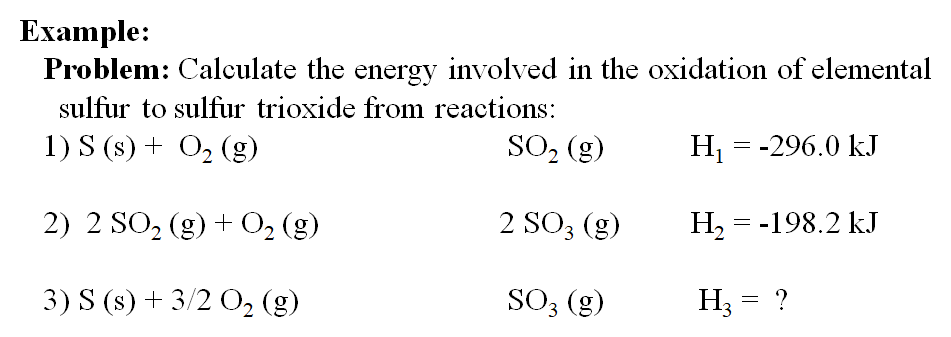

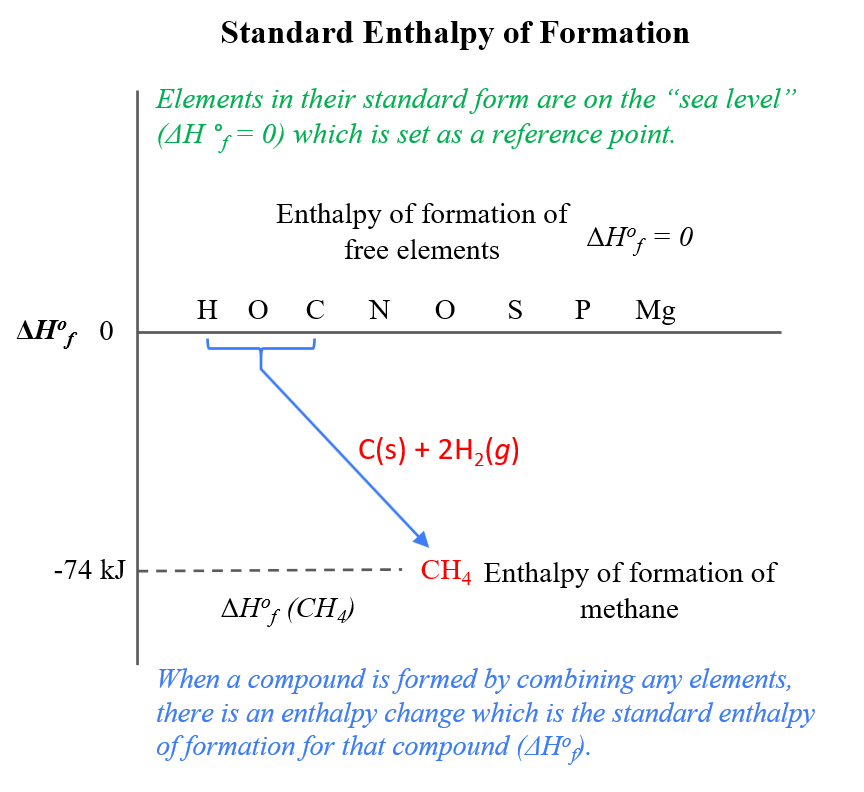



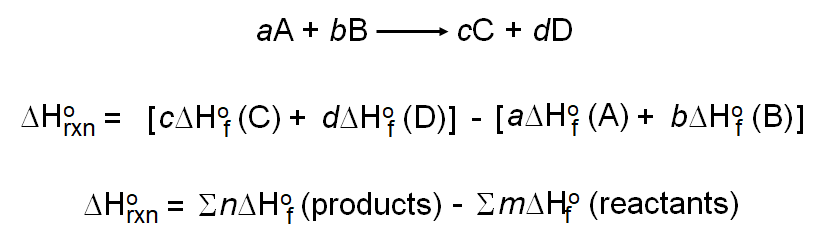





![5.1 Delta Hf and Delta Hc calculations [SL IB Chemistry] - YouTube 5.1 Delta Hf and Delta Hc calculations [SL IB Chemistry] - YouTube](https://i.ytimg.com/vi/zMksewz0cNU/maxresdefault.jpg)